All Categories
History






This section provides an overview for nickel metal hydride batteries as well as their applications and principles. Also, please take a look at the list of 6 nickel metal hydride battery manufacturers and their company rankings. Here are the top-ranked nickel metal hydride battery companies as of May, 2026: 1.Camelion Batterien GmbH, 2.DYNAMIS Batterien GmbH, 3.FDK CORPORATION.
Table of Contents
Categories Related to Nickel Metal Hydride Batteries

A nickel metal hydride battery is a type of rechargeable battery that can be charged and discharged, using a hydrogen storage alloy for the negative electrode and nickel hydroxide for the positive electrode.
Nickel metal hydride batteries are expensive because they use a hydrogen storage alloy instead of cadmium, but can be charged and discharged using a large current and have a large capacity per unit mass.
In addition, compared to other rechargeable batteries, nickel-metal hydride batteries have a relatively small memory effect (the voltage drop that occurs during discharge when batteries are repeatedly recharged without being fully discharged), and can be used repeatedly without performance degradation.
Nickel metal hydride batteries are used to take advantage of their high performance and long life, and are used in automotive batteries, notebook PCs, dry cell batteries, and other applications requiring high output and reliability.
In recent years, lithium-ion batteries, which do not have memory effect or self-discharge and have a larger capacity per unit mass and higher operating voltage, have come to be used.
Nickel metal hydride batteries consist of electrodes (positive electrode: nickel oxyhydroxide, negative electrode: hydrogen storage alloy), a separator made of olefin nonwoven fabric, and potassium hydroxide solution as electrolyte. In the case of a dry cell, the structure wound around these components is contained in a can.
During discharge of a nickel metal hydride battery, at the positive electrode, nickel oxyhydroxide receives electrons in the presence of water, producing nickel hydroxide and hydroxide ions. At the anode, hydrogen ions and electrons are released from the hydrogen storage alloy in the presence of hydroxide ions to produce water.
During discharge, the reaction proceeds in the opposite direction: at the cathode, hydroxide ions react with nickel hydroxide to produce nickel oxyhydroxide, which releases electrons. At the anode, hydrogen is adsorbed by supplying electrons.
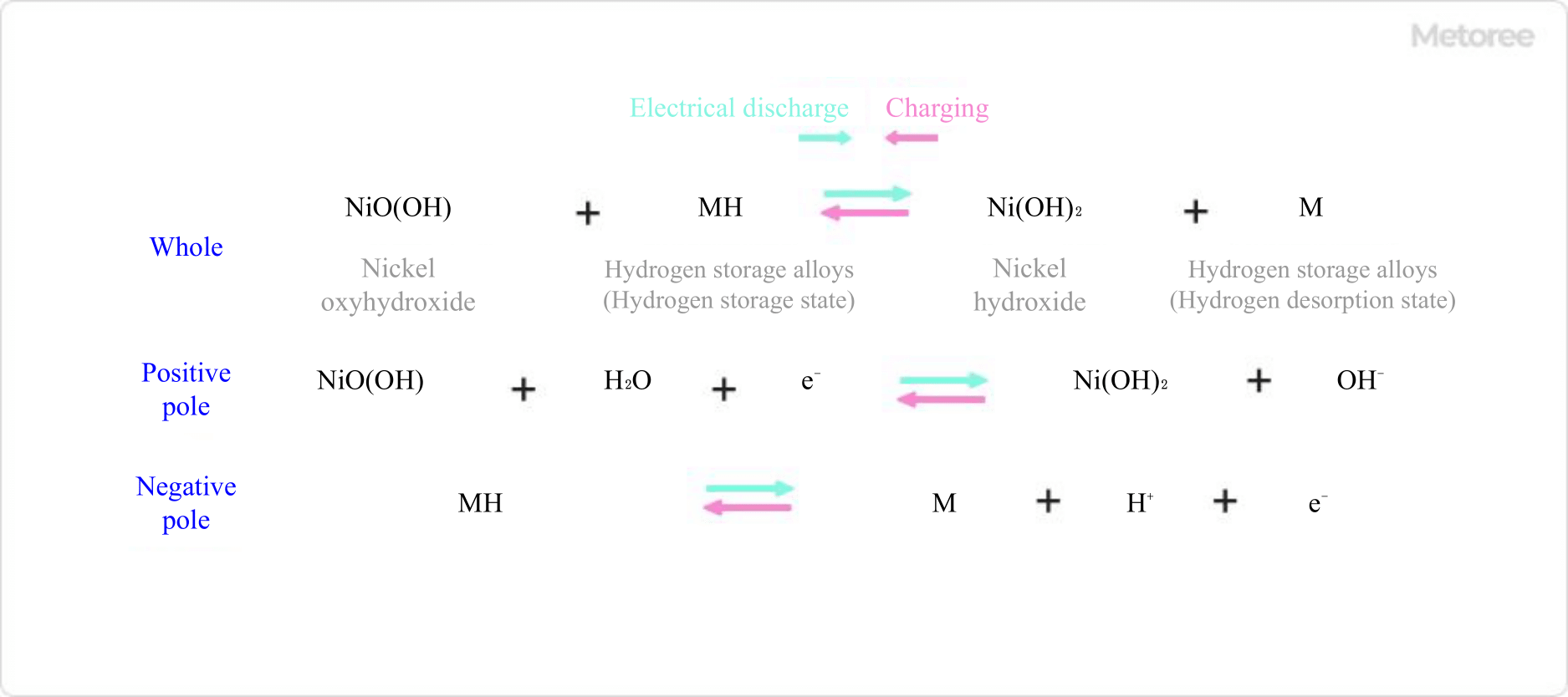
Figure 1. Charge-discharge reaction equation of a nickel-metal hydride battery
The charging and discharging of a nickel metal hydride battery occurs through a simple reaction that involves the adsorption of hydrogen and the production of water. For example, lead-acid batteries used in automobile batteries are charged and discharged through a precipitation-dissolution reaction of the electrodes, so repeated charging and discharging inevitably results in deterioration of the electrodes. A nickel metal hydride battery has no such degradation mode and can be used semi-permanently as long as the electrode itself does not deteriorate, making it a battery with a long service life.
Co-alloys have been mainly used for the negative electrode in the past in order to achieve high capacity, but there has been a move toward Co-free electrodes mainly due to cost considerations. However, the use of Co-fewer alloys has been progressing, mainly due to cost considerations. As for the cathode, nickel oxyhydroxide in the charged state is highly conductive, but nickel hydroxide in the discharged state is an insulator, which causes problems such as loss of electron paths during discharge. For this reason, cobalt oxyhydroxide or other materials are added to give conductivity.
1. Battery Characteristics
The nominal voltage of nickel metal hydride battery is 1.2V, which is the same as that of a nickel-cadmium battery. This is because the reactions used for charging and discharging are similar. Since the nominal voltage of lead-acid batteries is 2.0 V and the rated voltage of lithium-ion batteries is 3.7 V, they are relatively low voltage batteries. Since these batteries can easily carry a large current, they are used in equipment that requires high output, such as hybrid cars.
Nickel metal hydride batteries have a memory effect (the voltage of the battery drops as it is repeatedly recharged, resulting in a decrease in usable capacity and an inerting effect. Therefore, understanding the characteristics of the battery when using it will maximize its service life.
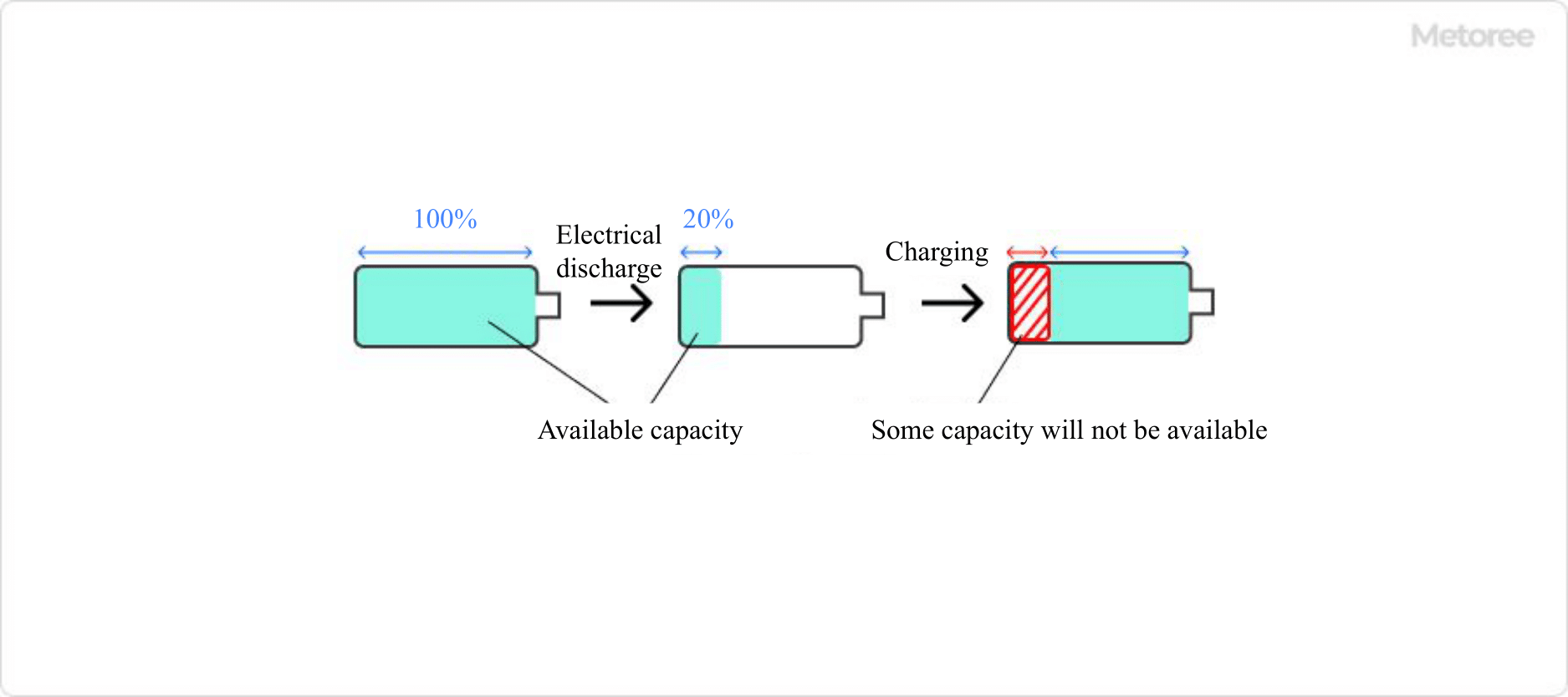
Figure 2. Memory effect
2. Safety
Basically, battery explosions and fires are caused by the ignition of organic solvents, which are electrolyte solvents, by sparks created by short circuits.
The electrolyte solvent in nickel metal hydride batteries is water, so even if a spark should occur, it will not ignite. Therefore, the current and voltage control mechanisms do not need to be designed as rigorously as in lithium-ion batteries, thus lowering the manufacturing cost. This low cost is one of the reasons why nickel metal hydride batteries are still widely used in industry.
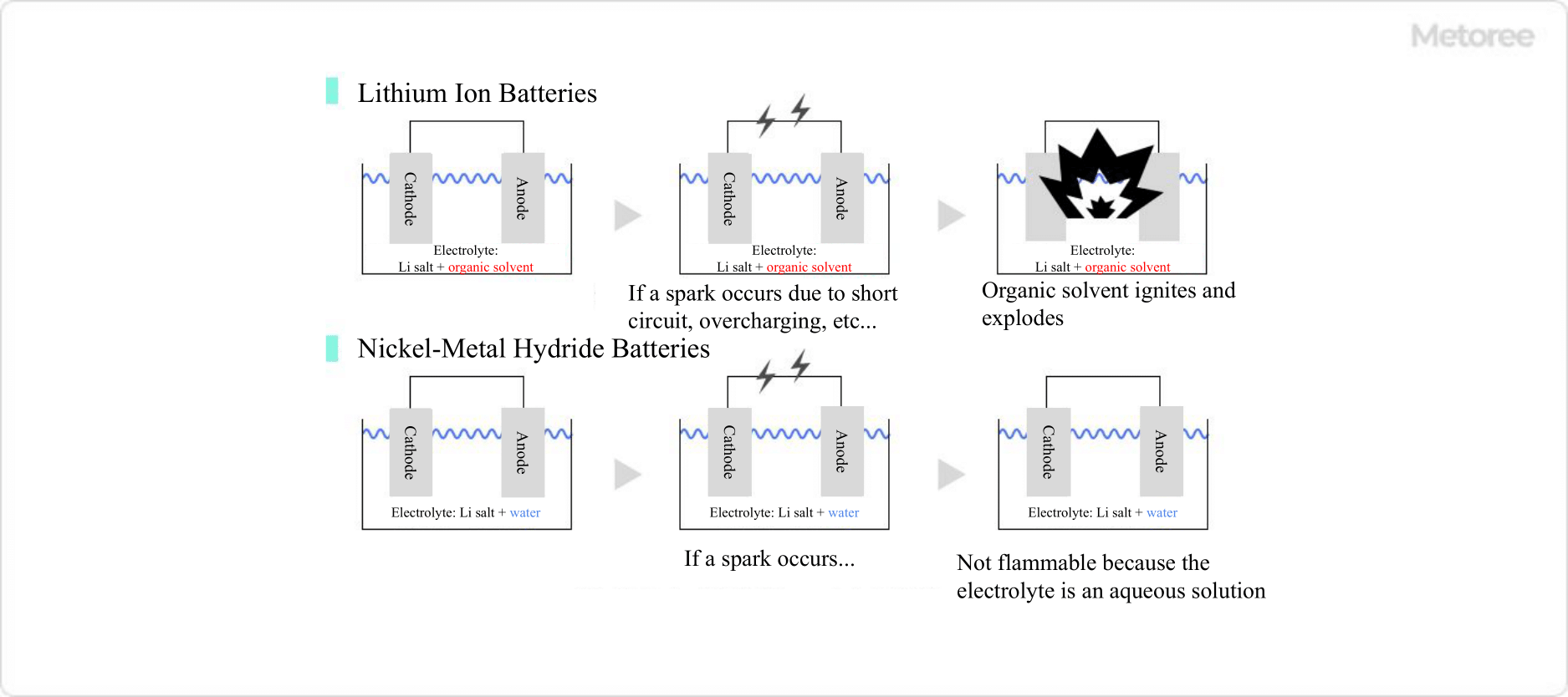
Figure 3. Comparison of lithium-ion and nickel-metal hydride batteries
3. Environmental Impact
Lithium-ion batteries, lead-acid batteries, and nickel-cadmium batteries contain hazardous substances with a high environmental impact (e.g., cadmium in nickel-cadmium batteries is a causative agent of Itai-itai disease, one of the four major pollution diseases). The electrolyte is also an environmentally friendly storage battery because it does not use organic solvents.
*Including some distributors, etc.
Sort by Features
Sort by Area

GmbH, founded in 2008 and based in Dettingen, Bayern, Germany, is a manufacturer and supplier of quality batteries and power storage solutions. The company's product portfolio includes a wide range of batteries, such as lead-acid batteries, lithium batteries, and gel batteries, designed for various applications like industrial, automotive, and renewable energy storage. Dynamis Batterien's advanced battery technologies ensure good power supply and efficient energy storage, meeting the diverse demands of industries worldwide. With an aim for sustainability, Dynamis Batterien aims to provide cutting-edge solutions that makes a greener and more efficient future.

Camelion Batterien GmbH was established in 1997 and is based in Berlin, Germany, as a manufacturer of power supply equipment. Its specialization is manufacturing alkaline batteries, rechargeable batteries, chargers, power banks, and flashlights. The company obtained several ISO certifications, including ISO 9001 and ISO 14000. It has partnered with companies such as Westinghouse and Shell. Its manufactured products are catered to the electronics, electrical appliances, automotive, and manufacturing fields across Germany.

FDK Corporation, founded in 1950 and headquartered in Tokyo, Japan, is a manufacturer and supplier of electronic components and batteries. Its diverse product range includes alkaline batteries for devices like toys and remote controls, Ni-MH batteries for eco-friendly rechargeable power, lithium batteries for medical devices and smart meters, carbon-zinc batteries for economical energy, and power supplies encompassing switching units and DC-DC converters. Furthermore, the company has several production plants located all over the globe, such as in Korea, Taiwan, Hong Kong, and Germany.

Jauch Group, established in 1954 and based in Villingen-Schwenningen, Germany, is a manufacturer and supplier of quartz crystals, crystal oscillators, and battery technology. The company’s products are used in various applications, including frequency control components for electronics and battery solutions for diverse industries. The company offers in-depth technical consulting, certification expertise, and fast product delivery. With production facilities in Germany and strategic partnerships in Asia, the company ensures the availability of frequency control products. Besides, the company provides new technology solutions and great customer support, catering to the evolving needs of its clients.

Toshiba Lifestyle Products & Services Corporation, located in Kawasaki City, Kanagawa, Japan, has been a manufacturer and distributor of household appliances since 1875. The company offers a diverse range of home appliances, including refrigerators, microwaves, vacuum cleaners, irons, and rice cookers, along with washing machines, and air conditioners. With seven domestic group companies and nine overseas bases in Asia, it also operates five manufacturing plants and four sales companies in Thailand, China, Vietnam, Malaysia, and Hong Kong. The company distributes its products and services to customers worldwide.

VB Power, established in 2008 by Mr. Zhang Wei and based in Dongguan City, Guangdong Province, China, is a supplier of power batteries. The company's product range encompasses NiMH battery series( SC, C, A, 2/3A, 4/5A) and Li-ion battery series (Lipo and LifePO4 battery) with high power and endurance to cryogenic temperature. The products are used in various industries, such as electric motors, cordless power tools, R/C cars and Hobbies, electric airsoft guns, AEG guns, and vacuum cleaners.
Ranking as of May 2026
Derivation Method| Rank | Company | Click Share |
|---|---|---|
| 1 | Camelion Batterien GmbH |
21.8%
|
| 2 | DYNAMIS Batterien GmbH |
19.1%
|
| 3 | FDK CORPORATION |
14.2%
|
| 4 | GPI International Limited |
12.1%
|
| 5 | Toshiba Lifestyle Products and Services Corporation |
9.7%
|
| 6 | Jauch Quartz |
8.7%
|
| 7 | VB power |
8.0%
|
| 8 | RPC Corporation |
6.3%
|
Derivation Method
The ranking is calculated based on the click share within the nickel metal hydride battery page as of May 2026. Click share is defined as the total number of clicks for all companies during the period divided by the number of clicks for each company.Number of Employees
Newly Established Company
Company with a History
*Including some distributors, etc.
| Country | Number of Companies | Share (%) |
|---|---|---|
 Germany
Germany
|
3 | 75.0% |
 Japan
Japan
|
1 | 25.0% |
62 products found
62 products
FDK Co., Ltd.
680+ people viewing
Last viewed: 1 hour ago
Features of FDK nickel metal hydride batteries ■Cathode material By using nickel hydroxide coated with highly conductive Co oxide, stable capacity ...
Nipron Co., Ltd.
570+ people viewing
Last viewed: 17 hours ago
◼︎Compatible power supply ・HNSP9-520 series ・eNSP3-450 series ・mNSP3-450 series This battery can be used with both nickel-metal hydride batteri...
Japan Lexel Co., Ltd.
490+ people viewing
Last viewed: 1 second ago
■Features ・High charging efficiency and rapid charging possible due to low internal resistance ・Capable of large charge/discharge like a capacito...
FDK Co., Ltd.
430+ people viewing
Last viewed: 1 hour ago
■Providing optimal assembled batteries and battery systems to meet your needs When batteries are used in devices, most of them are used in the form...
Japan Lexel Co., Ltd.
470+ people viewing
Last viewed: 1 minute ago
■Features ・Maintains a voltage of 1.0V or more when discharging at 20C, and 0.9V or more when discharging at 30C. ・Recommended for electric shave...
Japan Lexel Co., Ltd.
550+ people viewing
Our standard model
Japan Lexel Co., Ltd.
560+ people viewing
■Features - Approximately 90% remaining capacity after six months and 85% remaining capacity after one year ・Long number of cycles (approximately ...
IDEX Co., Ltd.
260+ people viewing
Last viewed: 11 hours ago
Connector, 24V charge/discharge 3P connector NR-203-RF (No.1 positive, No.2 negative, No.3 T terminal) [Maximum 10A], 24V discharge 2P connector NC...
Kanagawa Co., Ltd.
530+ people viewing
Last viewed: 1 day ago
Nickel-metal hydride batteries have approximately twice the capacity of nickel-cadmium storage batteries and have a voltage of 1.2V, making them co...
Nipron Co., Ltd.
580+ people viewing
Last viewed: 6 hours ago
◼︎Overview ・Built-in heater prevents capacity loss at low temperatures ・Battery pack status (charging/backup) can be output ・Backup time 3.5 min...
Kanagawa Co., Ltd.
540+ people viewing
Nickel-metal hydride batteries have approximately twice the capacity of nickel-cadmium storage batteries and have a voltage of 1.2V, making them co...
IDEX Co., Ltd.
230+ people viewing
Connector, 4P connector for charging only (No. 1 positive, No. 2 T terminal, No. 4 negative), 24V discharge 3P connector NR-203-RF (No. 1 positive,...
Kanagawa Co., Ltd.
660+ people viewing
Last viewed: 1 day ago
Nickel-metal hydride batteries have approximately twice the capacity of nickel-cadmium storage batteries and have a voltage of 1.2V, making them co...
Nipron Co., Ltd.
500+ people viewing
◼︎Overview ・Compatible with our 5-inch bay built-in lead-acid battery pack. Can be replaced with a nickel metal hydride battery during maintenance...
Nipron Co., Ltd.
450+ people viewing
Last viewed: 6 hours ago
◼︎Overview ・Compatible with our 5-inch bay built-in lead-acid battery pack. Can be replaced with a nickel metal hydride battery during maintenance...
Kanagawa Co., Ltd.
560+ people viewing
Nickel-metal hydride batteries have approximately twice the capacity of nickel-cadmium storage batteries and have a voltage of 1.2V, making them co...
Nipron Co., Ltd.
400+ people viewing
◼︎Features ・Large capacity lead acid battery ・Supports 450W peak output ・5 inch bay 2 unit size ・Small size that can be installed in 3.5 inch b...
Ueda Co., Ltd.
210+ people viewing
Last viewed: 1 day ago
12V, 3Ah
Ueda Co., Ltd.
220+ people viewing
Last viewed: 1 day ago
9.6V, 2Ah (small and lightweight type)
Ueda Co., Ltd.
220+ people viewing
Last viewed: 1 day ago
15.6V, 3Ah
Ueda Co., Ltd.
260+ people viewing
Last viewed: 1 day ago
■Product details information ・12V, 2Ah ・Approximately 1.6 times more work than Ni-Cd batteries
Ueda Co., Ltd.
270+ people viewing
Last viewed: 1 day ago
Nickel metal hydride 12V-3.0Ah
Ueda Co., Ltd.
260+ people viewing
Last viewed: 1 day ago
■Product details information ・7.2V, 2Ah ・Approximately 1.6 times more work than Ni-Cd batteries
Ueda Co., Ltd.
150+ people viewing
Last viewed: 7 hours ago
Cooling compatible battery
Ueda Co., Ltd.
330+ people viewing
Last viewed: 7 hours ago
■Applicable models REC-150F, REC-150FM, REC-1520FMH, REC-1520FH, REC-200M2H, REC-60G, REC-15G, FL-3012
Ueda Co., Ltd.
220+ people viewing
Last viewed: 1 day ago
Cooling compatible battery
Ueda Co., Ltd.
260+ people viewing
Last viewed: 15 hours ago
nickel metal hydride
Kessel Japan Co., Ltd.
350+ people viewing
Last viewed: 1 hour ago
Kessel Japan Co., Ltd.
330+ people viewing
Last viewed: 1 hour ago